Abstra t
Living organisms have evolved the ability to perceive and respond to light of different wavelengths within the visible spectrum by the generation of photoreceptor proteins. Recent studies revealed the participation of these proteins in the virulence of plant pathogenic bacteria. Pseudomonas syringae pv. tomato DC3000 (Pto) is responsible for the bacterial speck, which affects tomato crops. Pto genome contains two genes encoding red/far-red light photoreceptors (BphP1: PSPTO_1902 and BphP2: PSPTO_2652). This work demonstrates the participation of Pto phytochromes and light in the bacterial physiology and during the interaction with tomato plants. We found that Pto phytochromes are implicated in the control of some features related with the bacteria capability to enter into the plant apoplast and cause bacterial speck disease, such as motility, biofilm formation, adhesion and emulsification capability. Red light and bacteriophytochromes are important during the early colonization stage of tomato phyllosphere, affecting Pto virulence. In addition, the development of disease symptoms in infiltrated leaflets is affected by light, which may be the consequence of type-two secretion system regulation.







Similar content being viewed by others
References
Avila-Perez, M., Hellingwerf, K. J., & Kort, R. (2006). Blue light activates the sigmaB-dependent stress response of Bacillus subtilis via YtvA. Journal of Bacteriology, 188, 6411–6414.
Bai, Y., Rottwinkel, G., Feng, J., Liu, Y., & Lamparter, T. (2016). Bacteriophytochromes control conjugation in Agrobacterium fabrum. Journal of Photochemistry and Photobiology, B: Biology, 161, 192–199.
Beattie, G. A., & Lindow, S. E. (1995). The secret life of foliar bacterial pathogens on leaves. Annual Review of Phytopathology, 33, 145–172.
Beattie, G. A., & Lindow, S. E. (1999). Bacterial colonization of leaves: A spectrum of strategies. Phytopathology, 89, 353–359.
Bonomi, H. R., Toum, L., Sycz, G., Sieira, R., Toscani, A. M., Gudesblat, G. E., Leskow, F. C., Goldbaum, F. A., Vojnov, A. A., & Malamud, F. (2016). Xanthomonas campestris attenuates virulence by sensing light through a bacteriophytochrome photoreceptor. EMBO Reports. https://doi.org/10.15252/embr.201541691.
Branda, S. S., Vik, S., Friedman, L., & Kolter, R. (2005). Biofilms: The matrix revisited. Trends in Microbiology, 13, 20–26.
Buell, C. R., Joardar, V., Lindeberg, M., Selengut, J., Paulsen, I. T., Gwinn, M. L., Dodson, R. J., Deboy, R. T., Durkin, A. S., Kolonay, J. F., Madupu, R., Daugherty, S., Brinkac, L., Beanan, M. J., Haft, D. H., Nelson, W. C., Davidsen, T., Zafar, N., Zhou, L., Liu, J., Yuan, Q., Khouri, H., Fedorova, N., Tran, B., Russell, D., Berry, K., Utterback, T., Van Aken, S. E., Feldblyum, T. V., D'Ascenzo, M., Deng, W. L., Ramos, A. R., Alfano, J. R., Cartinhour, S., Chatterjee, A. K., Delaney, T. P., Lazarowitz, S. G., Martin, G. B., Schneider, D. J., Tang, X., Bender, C. L., White, O., Fraser, C. M., & Collmer, A. (2003). The complete genome sequence of the Arabidopsis and tomato pathogen Pseudomonas syringae pv. tomato DC3000. Proceedings of the National Academy of Sciences U S A, 100, 10181–10186.
Burch, A. Y., Zeisler, V., Yokota, K., Schreiber, L., & Lindow, S. E. (2014). The hygroscopic biosurfactant syringafactin produced by Pseudomonas syringae enhances fitness on leaf surfaces during fluctuating humidity. Environmental Microbiology, 16, 2086–2098.
Cameotra, S. S., & Bollag, J. M. (2003). Biosurfactant-enhanced bioremediation of polycyclic aromatic hydrocarbons. Critical Reviews in Environmental Science and Technology, 33, 111–126.
Choi, K. H., Kumar, A., & Schweizer, H. P. (2006). A 10-min method for preparation of highly electrocompetent Pseudomonas aeruginosa cells: Application for DNA fragment transfer between chromosomes and plasmid transformation. Journal of Microbiological Methods, 64, 391–397.
Costerton, J. W., Lewandowski, Z., Caldwell, D. E., Korber, D. R., & Lappin-Scott, H. M. (1995). Microbial biofilms. Annual Review of Microbiology, 49, 711–745.
Cunha, C. D., do Rosário, M., Rosado, A. S., & SGF, L. (2004). Serratia sp. SVGG16: A promising biosurfactant producer isolated from tropical soil during growth with ethanol-blended gasoline. Process Biochemistry, 39, 2277–2282.
Dow, J. M., Crossman, L., Findlay, K., He, Y. Q., Feng, J. X., & Tang, J. L. (2003). Biofilm dispersal in Xanthomonas campestris is controlled by cell-cell signaling and is required for full virulence to plants. Proceedings of the National Academy of Sciences U S A, 100, 10995–11000.
Dunger, G., Relling, V. M., Tondo, M. L., Barreras, M., Ielpi, L., Orellano, E. G., & Ottado, J. (2007). Xanthan is not essential for pathogenicity in citrus canker but contributes to Xanthomonas epiphytic survival. Archives of Microbiology, 188, 127–135.
Ficarra, F.A., Grandellis, C., Galván, E.M., Ielpi, L., Feil, R., Lunn, J.E., Gottig, N., & Ottado, J. (2016). Xanthomonas citri ssp. citri requires the outer membrane porin OprB for maximal virulence and biofilm formation. Molecular Plant Pathology, 18, 720–733.
Fixen, K. R., Baker, A. W., Stojkovic, E. A., Beatty, J. T., & Harwood, C. S. (2014). Apo-bacteriophytochromes modulate bacterial photosynthesis in response to low light. Proceedings of the National Academy of Sciences U S A, 111, 237–244.
Gomelsky, M., & Hoff, W. D. (2011). Light helps bacteria make important lifestyle decisions. Trends in Microbiology, 19, 441–448.
Gottig, N., Garavaglia, B. S., Garofalo, C. G., Orellano, E. G., & Ottado, J. (2009). A filamentous hemagglutinin-like protein of Xanthomonas axonopodis pv. citri, the phytopathogen responsible for citrus canker, is involved in bacterial virulence. PLoS One, 4, e4358.
Hockett, K. L., Burch, A. Y., & Lindow, S. E. (2013). Thermo-regulation of genes mediating motility and plant interactions in Pseudomonas syringae. PLoS One, 8, e59850.
Jha, G., Rajeshwari, R., & Sonti, R. V. (2005). Bacterial type two secretion system secreted proteins: Double-edged swords for plant pathogens. Molecular Plant-Microbe Interactions Journal, 18, 891–898.
Karatan, E., & Watnick, P. (2009). Signals, regulatory networks, and materials that build and break bacterial biofilms. Microbiology and Molecular Biology Reviews, 73, 310–347.
Kearns, D. B. (2010). A field guide to bacterial swarming motility. Nature Reviews Microbiology, 8, 634–644.
King, E. O., Ward, M. K., & Raney, D. E. (1954). Two simple media for the demonstration of pyocyanin and fluorescein. The Journal of Laboratory and Clinical Medicine, 44, 301–307.
Kovach, M. E., Elzer, P. H., Hill, D. S., Robertson, G. T., Farris, M. A., Roop, R. M., & Peterson, K. M. (1995). Four new derivatives of the broad-host-range cloning vector pBBR1MCS, carrying different antibiotic-resistance cassettes. Gene, 166, 175–176.
Kraiselburd, I., Alet, A. I., Tondo, M. L., Petrocelli, S., Daurelio, L. D., Monzon, J., Ruiz, O. A., Losi, A., & Orellano, E. G. (2012). A LOV protein modulates the physiological attributes of Xanthomonas axonopodis pv. citri relevant for host plant colonization. PLoS One, 7, e38226.
Kraiselburd, I., Moyano, L., Carrau, A., Tano, J., & Orellano, E. G. (2017). Bacterial photosensory proteins and their role in plant–pathogen interactions. Photochemistry and Photobiology, 93, 666–674.
Lee, B., Haagensen, J. A., Ciofu, O., Andersen, J. B., Hoiby, N., & Molin, S. (2005). Heterogeneity of biofilms formed by nonmucoid Pseudomonas aeruginosa isolates from patients with cystic fibrosis. Journal of Clinical Microbiology, 43, 5247–5255.
McGrane, R., & Beattie, G. A. (2017). Pseudomonas syringae pv. syringae B728a regulates multiple stages of plant colonization via the bacteriophytochrome BphP1. mBio, 8, e01178-17.
Moriconi, V., Sellaro, R., Yub, N., Soto, G., Rugnone, M., Shah, R., Pathak, G. P., Gartner, W., & Casal, J. J. (2013). LOV-domain photoreceptor, encoded in a genomic island, attenuates the virulence of Pseudomonas syringae in light-exposed Arabidopsis leaves. Plant Journal, 76, 322–331.
Mussi, M. A., Gaddy, J. A., Cabruja, M., Arivett, B. A., Viale, A. M., Rasia, R., & Actis, L. A. (2010). The opportunistic human pathogen Acinetobacter baumannii senses and responds to light. Journal of Bacteriology, 192, 6336–6345.
Nagendran, R., & Lee, Y. H. (2015). Green and red light reduces the disease severity by Pseudomonas cichorii JBC1 in tomato plants via upregulation of defense-related gene expression. Phytopathology, 105, 412–418.
Nagendran, R., & Lee, Y. H. (2018). Effects of green light on the gene expression and virulence of the plant pathogen Pseudomonas cichorii JBC1. European Journal of Plant Pathology, 150, 223–236.
Nickzad, A., & Dèziel, E. (2014). The involvement of rhamnolipids in microbial cell adhesion and biofilm development -an approach for control? Letters in Applied Microbiology, 58, 447–453.
Nitschke, M., & Pastore, G. M. (2006). Production and properties of a surfactant obtained from Bacillus subtilis grown on cassava wastewater. Bioresource Technology, 97, 336–341.
Njimona, I., Yang, R., & Lamparter, T. (2014). Temperature effects on bacterial phytochrome. PLoS One, 9, e109794.
Petrocelli, S., Tondo, M.L., Daurelio, .LD., & Orellano, E. G. (2012). Modifications of Xanthomonas axonopodis pv. citri lipopolysaccharide affect the basal response and the virulence process during citrus canker. PLoS One, 7, e40051.
Preston, G. M. (2000). Pseudomonas syringae pv. tomato: The right pathogen, of the right plant, at the right time. Molecular Plant Pathology, 1, 263–275.
Purcell, E. B., McDonald, C. A., Palfey, B. A., & Crosson, S. (2010). An analysis of the solution structure and signaling mechanism of LovK, a sensor histidine kinase integrating light and redox signals. Biochemistry, 49, 6761–6770.
Ricci, A., Dramis, L., Shah, R., Gartner, W., & Losi, A. (2015). Visualizing the relevance of bacterial blue- and red-light receptors during plant-pathogen interaction. Environmental Microbiology Reports, 7, 795–802.
Rigano, L. A., Siciliano, F., Enrique, R., Sendin, L., Filippone, P., Torres, P. S., Questa, J., Dow, J. M., Castagnaro, A. P., Vojnov, A. A., & Marano, M. R. (2007). Biofilm formation, epiphytic fitness, and canker development in Xanthomonas axonopodis pv. citri. Molecular Plant-Microbe Interactions Journal, 20, 1222–1230.
Río Alvarez, I., Rodriguez-Herva, J. J., Martinez, P. M., Gonzalez-Melendi, P., Garcia-Casado, G., Rodriguez-Palenzuela, P., & Lopez-Solanilla, E. (2014). Light regulates motility, attachment and virulence in the plant pathogen Pseudomonas syringae pv tomato DC3000. Environmental Microbiology, 16, 2072–2085.
Rockwell, N. C., Su, Y. S., & Lagarias, J. C. (2006). Phytochrome structure and signaling mechanisms. Annual Review of Plant Biology, 57, 837–858.
Roden, L. C., & Ingle, R. A. (2009). Lights, rhythms, infection: The role of light and the circadian clock in determining the outcome of plant-pathogen interactions. Plant Cell, 21, 2546–2552.
Sambrook, J., Fritsch, E. F., & Maniatis, T. (1989). Molecular cloning. A laboratory manual (1st ed.). New York: Cold Spring Harbor Laboratory Press.
Santamaría-Hernando, S., Rodríguez-Herva, J. J., Martínez-García, P. M., Río-Alvarez, I., González-Melendi, P., Jaime Zamorano, J., Tapia, C., Rodríguez-Palenzuela, P., & López-Solanilla, E. (2018). Pseudomonas syringae pv. tomato exploits light signals to optimize virulence and colonization of leaves. Environmental Microbiology, 20, 4261–4280.
Shah, R., Pathak, G., Drepper, T., & Gartner, W. (2016). Selective photoreceptor gene knock-out reveals a regulatory role for the growth behavior of Pseudomonas syringae. Photochemistry and Photobiology, 92, 571–578.
Smibert, R. M. & Krieg, N. R. (1994). Phenotypic characterization. In P. Gerhardt, R. G. E. Murray, W. A. Wood & N. R. Krieg (Eds.), Methods for general and molecular bacteriology (pp. 611–651). Washington, DC: American Society for Microbiology.
Stoodley, P., Sauer, K., Davies, D. G., & Costerton, J. W. (2002). Biofilms as complex differentiated communities. Annual Review of Microbiology, 56, 187–209.
Tondo, M.L., Delprato, M.L., Kraiselburd, I., Fernández Zenoff, M.V., Farías, M.E., & Orellano, E. G. (2016). KatG, the bifunctional catalase of Xanthomonas citri subsp. citri, responds to hydrogen peroxide and contributes to epiphytic survival on citrus leaves. PLoS One, 11, e0151657.
Thomas, P., & Upreti, R. (2014). Significant effects due to peptone in Kelman medium on colony characteristics and virulence of Ralstonia solanacearum in tomato. The Open Microbiology Journal, 8, 95–114.
Uzoigwe, C., Burgess, J. G., Ennis, C. J., & Rahman, P. K. S. M. (2015). Bioemulsifiers are not biosurfactants and require different screening approaches. Frontiers in Microbiology, 6, 245. https://doi.org/10.3389/fmicb.2015.00245.
van der Horst, M. A., Key, J., & Hellingwerf, K. J. (2007). Photosensing in chemotrophic, non-phototrophic bacteria: Let there be light sensing too. Trends in Microbiology, 15, 554–562.
Wu, L., McGrane, R. S., & Beattie, G. A. (2013). Light regulation of swarming motility in Pseudomonas syringae integrates signaling pathways mediated by a bacteriophytochrome and a LOV protein. mBio, 4, e00334-13.
Xin, X. F., & He, S. Y. (2013). Pseudomonas syringae pv. tomato DC3000: A model pathogen for probing disease susceptibility and hormone signaling in plants. Annual Review of Phytopathology, 51, 473–498.
Xu, J., Plat, T. G., & Fuqua, C. (2012). Regulatory linkages between flagella and surfactant during swarming behavior: Lubricating the flagellar propeller? Journal of Bacteriology, 194, 1283–1286.
Yang, Y. X., Wang, M. M., Yin, Y. L., Onac, E., Zhou, G. F., Peng, S., Xia, X. J., Shi, K., Yu, J. Q., & Zhou, Y. H. (2015). RNA-seq analysis reveals the role of red light in resistance against Pseudomonas syringae pv. tomato DC3000 in tomato plants. BMC Genomics, 16, 120.
Acknowledgments
We thank, Rodrigo Vena (IBR-CONICET) for microscopic analyses, the Statistics Department (Cátedra de Estadística Facultad de Ciencias Bioquímicas y Farmacéuticas, Universidad Nacional de Rosario) for their assistance in the statistical analysis of the data and the staff from the English Department of the Facultad de Ciencias Bioquímicas y Farmacéuticas (UNR) for the language correction of the manuscript. The anti-flagellin antibodies were kindly provided by the laboratory of Dr. Eleonora García Véscovi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This research article is not submitted elsewhere for publication and this manuscript complies with the Ethical Rules applicable for this journal.
Competing interests
The authors declare that they have no competing interest.
Human and animal studies
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Fig. S1
Adhesion and biofilm formation by P. syringae pv. tomato DC3000 exposed to white light, red light and dark conditions. (A) Representative photograph of Pto WT, bphP1::Spr, cbphP1::Spr, bphP2::Gnr and cbphP2::Gnr adhered to the abaxial leaf surface and stained with Crystal violet dye. KB medium was taken as control. The order of inoculation is indicated at the left side of the panel. Dashed lines in the leaves indicate the inoculated area. (B) Representative photographs of biofilm formation assay. Pto strains were statically grown on glass flasks for 24 h at 28 °C. Biofilm formation levels on the air-liquid-glass interface were determined by Crystal violet staining. (D) Representative photographs of green fluorescent protein (GFP)-labeled bacteria were visualized under confocal laser scanning microscopy (CLSM) after three days of bacterial growth. Scale bars 50 μm. (PNG 2087 kb)
Fig S2
Biosurfactant production of P. syringae pv. tomato DC3000 exposed to white light, red light and dark conditions. Representative photograph of Pto WT, bphP1::Spr, cbphP1::Spr, bphP2::Gnr and cbphP2::Gnr biosurfactant production. (PNG 1948 kb)
Fig. S3
Effect of phytochrome genes disruption of P. syringae pv. tomato DC3000 and light on the epiphytic fitness. (A) Representative photograph of disease symptoms on tomato leaflets inoculated by dipping with Pto WT, bphP1::Spr, cbphP1::Spr, bphP2::Gnr and cbphP2::Gnr strains and exposed to white light, red light and darkness. (PNG 1130 kb)
Fig. S4
Bacterial growth curves in KB liquid medium. Growth curves of Pto WT, bphP1::Spr, cbphP1::Spr, bphP2::Gnr and cbphP2::Gnr strains in fresh KB medium considering the optical density at 600 nm (OD600) under (A) white light, (B) red light and (C) darkness, and the CFU/mL under (D) white light, (E) red light and (F) darkness, as a function of time. Data are represented as the mean ± standard error of two independent experiments. (PNG 407 kb)
Fig. S5
Expression of bacteriophytochrome genes in WT and complemented strains. (A) Amplified products of the phytochrome genes by semiquantitative RT-PCR using RNA preparation from WT and complemented strains. As a control for constitutive bacterial expression a fragment of 16S rRNA was simultaneously amplified. (PNG 45 kb)
Appendix S1
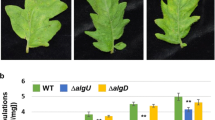
. Statistical analysis for Figs. 1B, 5B and 6B. (DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Moyano, L., Carrau, A., Petrocelli, S. et al. Bacteriophytochromes from Pseudomonas syringae pv. tomato DC3000 modulate the early stages of plant colonization during bacterial speck disease. Eur J Plant Pathol 156, 695–712 (2020). https://doi.org/10.1007/s10658-019-01918-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-019-01918-5